
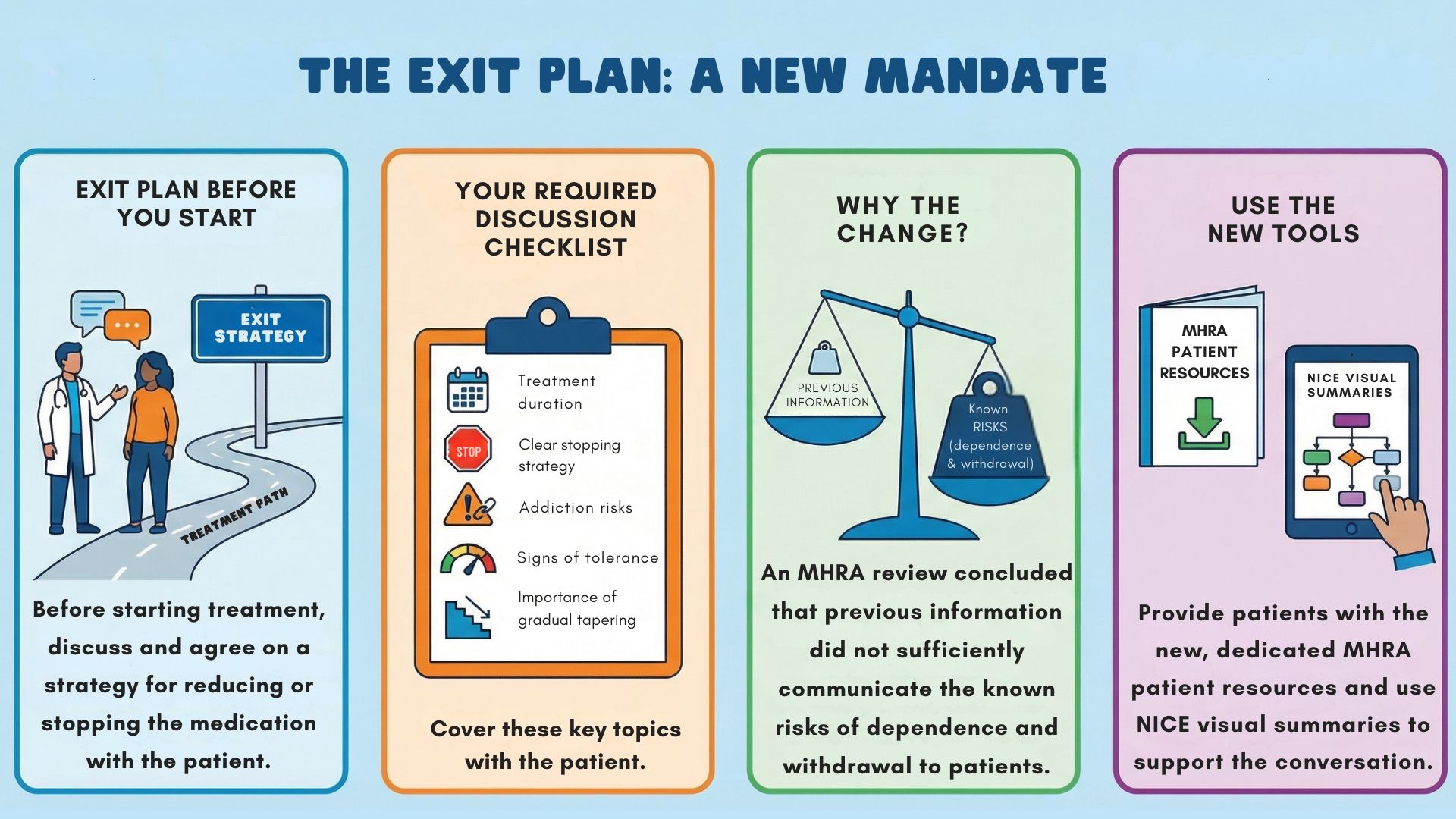
On January 8th, the MHRA published new guidance following a review by the Commission on Human Medicines. The message: before starting treatment with these medications, clinicians should hold a discussion with patients to put in place a strategy for reducing or ending treatment.
But Why Now?
The MHRA reviewed Yellow Card data, GP databases, reports from other regulators, and crucially, accounts from patient groups. Their conclusion? Current product information didn’t adequately communicate the risks. Patients weren’t being properly informed before starting treatment, and the conversation about stopping wasn’t happening early enough, or sometimes at all.
What the guidance says:
Download the infographic
The guidance is clear - these need to be meaningful discussions, not box-ticking.
Interestingly, while it emphasizes having the conversation, it doesn’t explicitly mandate specific documentation; however, good practice suggests noting what was discussed and agreed. The MHRA is particularly concerned about tolerance. If patients say the medication isn’t working as well and they need more, that’s the moment to explore alternatives, not simply increase the dose.
Beyond the guidance, there are practical changes rolling out over the coming months.
Updated warnings on product packaging
Strengthened SPCs and Patient Information Leaflets
New MHRA patient resources
The MHRA review concluded that current information “did not sufficiently communicate the extent of the known risks.” This update attempts to fix that by ensuring the conversation happens upfront. The guidance is clear: have the conversation early. Make the exit plan before you start. Whether this will meaningfully reduce long-term dependency remains to be seen.

